Atomic spectra meaning8/28/2023  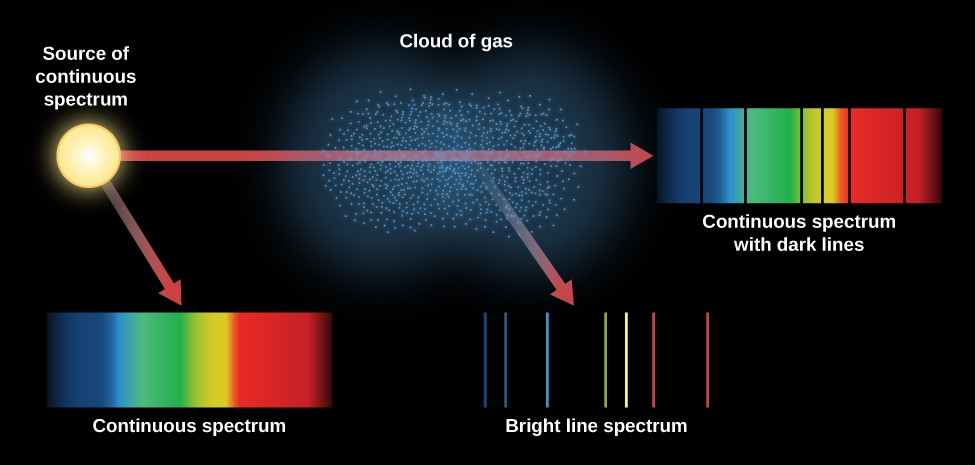
Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron.\) for a short animation. Molecular spectra are observed when a molecule undergoes the absorption or emission of electromagnetic radiation with a resulting increase or decrease in energy. atomic spectrum synonyms, atomic spectrum pronunciation, atomic spectrum translation, English dictionary definition of atomic spectrum. The compilation includes data for the neutral and singly-ionized atoms of all elements hydrogen through einsteinium ( Z 1-99). It ranges from the longest radio waves to the shortest X-rays and gamma rays. Introduction This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily accessible format. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. Spectrum: Definition The spectrum is a continuous range of electromagnetic radiation waves. Excited state: to be in a higher energy level. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Emission spectrum (or atomic spectrum): The unique pattern of light given off by an element when it is given energy Ground state: to be in the lowest energy level possible. Absorption or Dark Line Three types of spectra: continuous, emission line and absorption. 
Figure : An absorption spectrum of anthracene. Examples of absorption and fluorescence spectra are shown in Figures and. When hydrogen gas is placed into a tube and electric current passed through it, the color of emitted light is pink. Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. Alkalis and Alkali-like Spectra In the central field approximation there exists no angular-momentum coupling between a closed subshell and an electron outside the subshell, since the net spin and orbital angular momenta of the subshell are both zero. A spectrum is a graph that shows the intensity of radiation at different wavelengths or the response of the atomic or molecular system to different wavelengths of the radiation. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
